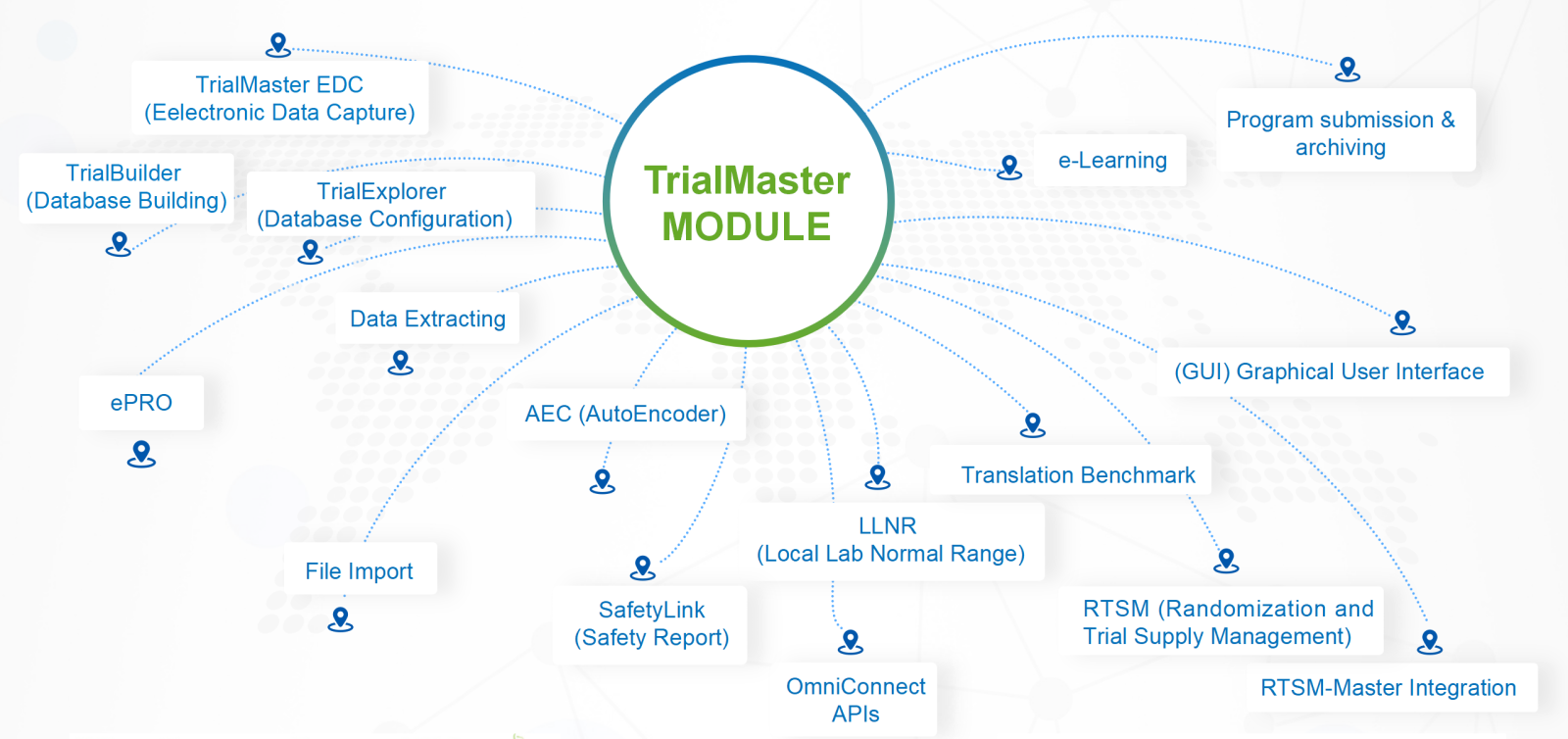
EN
Easy for user access and use: the system adapts Liquid Layout screen design. It can automatically adjust the layout proportion according to the equipment, ensuring the users have friendly experience of operation across different devices.
Real-time reflect discrepancy: the system provide the real-time guidance feedback to user who entering data. It can reduce data errors and avoid expensive data clean cost.
Support multi-language requirements: the system can adopt different nations and area needs via translation benchmark, provide multi-lingual interface that fits their habits. Users can switch language as needed.